What about the acetate ion? If it hydrolyzes, it will take an H + from a water molecule:Ĭ 2H 3O 2 −(aq) + H 2O → HC 2H 3O 2 + OH −(aq)ĭoes this happen? Yes, it does. We already know that the Na + ion won’t affect the acidity of the solution. Things change, however, when we consider a salt like NaC 2H 3O 2. Because neither ion in NaCl affects the acidity or basicity of the solution, NaCl is an example of a neutral salt. The net result? There is no change, so there is no effect on the acidity or basicity of the solution from the Cl −(aq) ion. The free H +(aq) ion reacts with the OH −(aq) ion to remake a water molecule: 
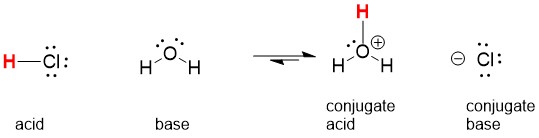
However, HCl is a strong acid, which means that it is 100% ionized in solution: What about the Cl − ion? Will it hydrolyze? If it does, it will take an H + ion from a water molecule: The net result? There is no change, so there is no effect on the acidity or basicity of the solution from the Na +(aq) ion. The free OH −(aq) ion reacts with the H +(aq) ion to remake a water molecule: However, NaOH is a strong base, which means that it is 100% ionized in solution: Will the Na +(aq) ion hydrolyze? If it does, it will interact with the OH − ion to make NaOH: When it dissolves in an aqueous solution, it separates into Na + ions and Cl − ions: The general rule is that salts with ions that are part of strong acids or bases will not hydrolyze, while salts with ions that are part of weak acids or bases will hydrolyze.Ĭonsider NaCl. It does not proceed 100% to products because hydrazoic acid is not a strong acid.Ĭertain salts will also affect the acidity or basicity of aqueous solutions because some of the ions will undergo hydrolysis, just like NH 3 does to make a basic solution. Write the balanced chemical equation for the dissociation of hydrazoic acid (HN 3) and indicate whether it proceeds 100% to products or not. When an ionic compound dissolves, it separates into its constituent ions:īecause Ca(OH) 2 is listed in Table 12.2 “Strong Acids and Bases”, this reaction proceeds 100% to products. This is an ionic compound of Ca 2+ ions and OH − ions. Write the balanced chemical equation for the dissociation of Ca(OH) 2 and indicate whether it proceeds 100% to products or not. 
So a base based on some other mechanism, such as NH 3 (which does not contain OH − ions as part of its formula), will be a weak base. There are very few strong bases (see Table 12.2 “Strong Acids and Bases”) any base not listed is a weak base. 
If it is less than 100% ionized in solution, it is a weak base. The issue is similar with bases: a strong base is a base that is 100% ionized in solution. It may be 1% ionized or 99% ionized, but it is still classified as a weak acid. If an acid is not listed here, it is a weak acid. HC 2H 3O 2→ H +(aq) + C 2H 3O 2 −(aq) (~5%)īecause this reaction does not go 100% to completion, it is more appropriate to write it as an equilibrium:Īs it turns out, there are very few strong acids, which are given in Table 12.2 “Strong Acids and Bases”. If it does not dissociate 100%, it is a weak acid. When HCl is dissolved in H 2O, it completely dissociates into H +(aq) and Cl −(aq) ions all the HCl molecules become ions:Īny acid that dissociates 100% into ions is called a strong acid. However, acids can be very different in a very important way.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
